Phone: (858) 882-2800
The CFTR gene encodes the cystic fibrosis transmembrane conductance regulator, a chloride channel expressed in epithelial cells of the lungs, pancreas, gastrointestinal tract, sweat glands, and reproductive system. Proper CFTR function regulates salt and water transport across cell membranes. When pathogenic variants disrupt this function, thickened secretions accumulate, leading to the multi-system manifestations of cystic fibrosis (CF).4
Cystic fibrosis is one of the most common autosomal recessive genetic conditions in individuals of Northern European ancestry, with a prevalence of approximately 1 in 2,500–3,300 live births, though it affects individuals across all genetic ancestries.2

Identifies individuals who carry a single pathogenic CFTR variant and may be at risk of having a child with CF if both partners are carriers.

Aims to identify affected infants shortly after birth to enable early intervention and referral.
CF was the first genetic condition for which pan-ethnic carrier screening was broadly recommended. In 2001, laboratory standards were introduced outlining a core set of CFTR variants suitable for population-based testing.3 This was followed in 2004 by refinement to a 23-variant minimum panel, widely known as the ACMG-23 panel.2
At the time, variant selection reflected available epidemiologic data and technical capabilities. However, subsequent research demonstrated that small, fixed panels do not provide equivalent carrier detection across diverse populations. Variant frequency varies significantly by ancestry, and limited panels can underrepresent pathogenic variants more common outside Northern European populations.2
Recognizing these disparities, the American College of Medical Genetics and Genomics (ACMG) updated its recommendations in 2023, expanding the minimum recommended CFTR carrier screening variant list to 100 well-established pathogenic variants.1 This update emphasized improving variant representation across biogeographic ancestries while maintaining rigorous pathogenicity criteria.1
Recent analysis of commercially available CFTR carrier screening assays has shown that not all panels fully align with the updated ACMG 100-variant recommendations.6 For laboratories performing carrier screening, periodic review of variant content is essential to ensure guideline alignment.
Carrier frequency for CFTR variants varies significantly by ancestry.2 The original 23-variant panel was developed based on available data that was disproportionately reflective of Northern European populations.2,3 As a result, estimated detection rates differed substantially across ancestral groups.
Based on cumulative CFTR variant frequencies reported by ancestry, the estimated detection rate of the previous ACMG-recommended 23-variant panel varied considerably.
These differences highlight how limited variant sets can underrepresent pathogenic variants more common in certain populations. As the U.S. population becomes increasingly diverse, broader CFTR variant coverage plays an important role in reducing ancestry-based disparities in detection.
The transition from the 23-variant panel2 to the ACMG-recommended 100-variant set¹ reflects advances in genomic databases, improved variant curation, and expanded population representation. By incorporating a larger number of well-established pathogenic variants, laboratories can improve detection sensitivity across ancestries while maintaining alignment with current professional standards.¹
For both carrier screening and newborn screening laboratories, expanded variant coverage should be paired with clearly defined interpretation frameworks and transparent reporting practices.4
Genetic counseling guidance further emphasizes the importance of communicating residual risk and variant meaning appropriately in reproductive contexts.5

Figure 1: Estimated detection rate of previous ACMG-recommended 23-variant panel
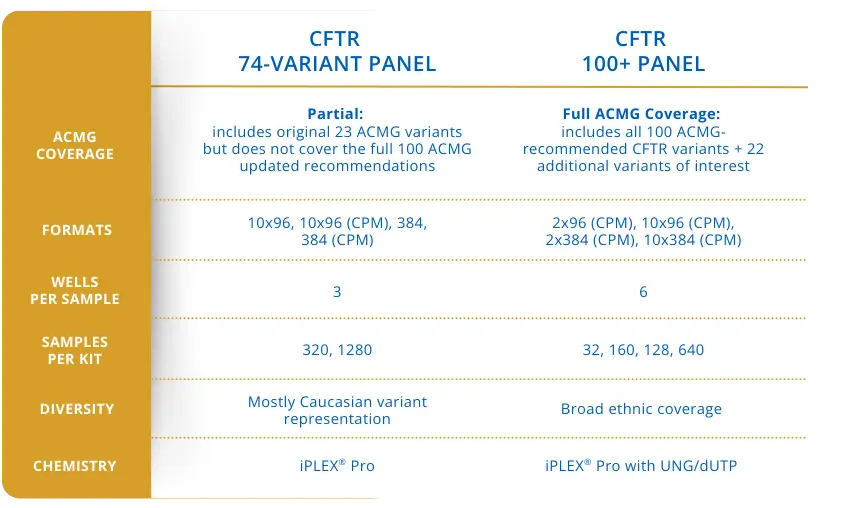
Figure 2: Comparison table of Agena Bioscience CFTR 74-variant panel versus the new CFTR 100+ Panel
The CFTR 100+ Panel, developed for use on the MassARRAY® System, was designed to support expanded CFTR carrier screening in research and public health settings. The panel includes all CFTR variants recommended by the ACMG for carrier screening, along with additional variants of interest informed by emerging evidence and laboratory input. By leveraging a multiplexed genotyping workflow, the assay enables expanded CFTR variant coverage while maintaining efficiency and reproducibility in high-volume laboratory environments.
This approach allows public health and newborn screening laboratories to align with current carrier screening expectations while retaining the flexibility needed to adapt as evidence and guidance continue to evolve.
References
1. Deignan JL, Gregg AR, Grody WW, et al. Updated recommendations for CFTR carrier screening: A position statement of the American College of Medical Genetics and Genomics (ACMG). Genetics in Medicine. 2023.2. Watson MS, Cutting GR, Desnick RJ, et al. Cystic fibrosis population carrier screening: 2004 revision of American College of Medical Genetics mutation panel. Genetics in Medicine. 2004. 3. Grody WW, Cutting GR, Klinger KW, et al. Laboratory standards and guidelines for population-based cystic fibrosis carrier screening. Genetics in Medicine. 2001. 4. Deignan JL, Astbury C, Cutting GR, et al. CFTR variant testing: a technical standard of the American College of Medical Genetics and Genomics (ACMG). Genetics in Medicine. 2020. 5. Langfelder-Schwind E, et al. Cystic fibrosis prenatal screening in genetic counseling practice: recommendations of the National Society of Genetic Counselors. Journal of Genetic Counseling. 2005. 6. Tidwell T, Gregg AR, Grody WW,et al. Commercial cystic fibrosis carrier screening tests and coverage of the ACMG 100 recommended variants. Genetics in Medicine Open. 2024.
See how the CFTR 100+ Panel delivers validated accuracy, robustness, and reproducibility across multiple laboratories using the MassARRAY System.
Includes analytical performance data, concordance results, and real-world workflow validation.
Download White Paper